Overview
Antimicrobial resistance (AMR) is a global issue with increasingly severe consequences for the world economy and human health. Vaccines reduce AMR by preventing infectious diseases, but the benefits of vaccination campaigns in improving health and economic outcomes have yet to be extensively quantified. ARVac, a consortium of One Health Trust (founded as CDDEP), Yale, Berkeley, Imperial College London, and Princeton, brings together experts in infectious disease modeling to assess the health and economic benefits of potential, new, and current vaccines targeting pathogens of public health relevance.
Vaccines and AMR
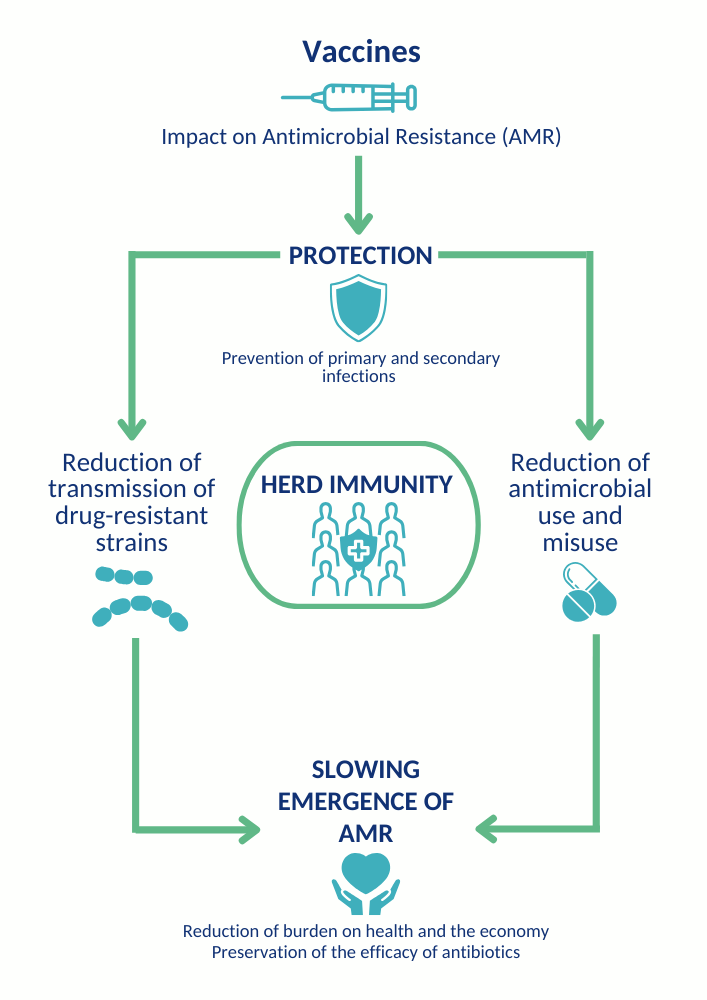
Many vaccines, both those in current use and those in the pipeline, have the potential to reduce the AMR disease burden, and thereby save lives, via two mechanisms:
- A lower overall burden of infections reduces the transmission of resistant and susceptible pathogens.
- Fewer infections reduce the need for antimicrobials, thereby alleviating the selection pressure for resistant pathogenic strains. Ultimately, the drop in resistant cases leads to fewer untreatable infections and more lives saved.
Synergizing Immunization and AMR Mitigation Strategies
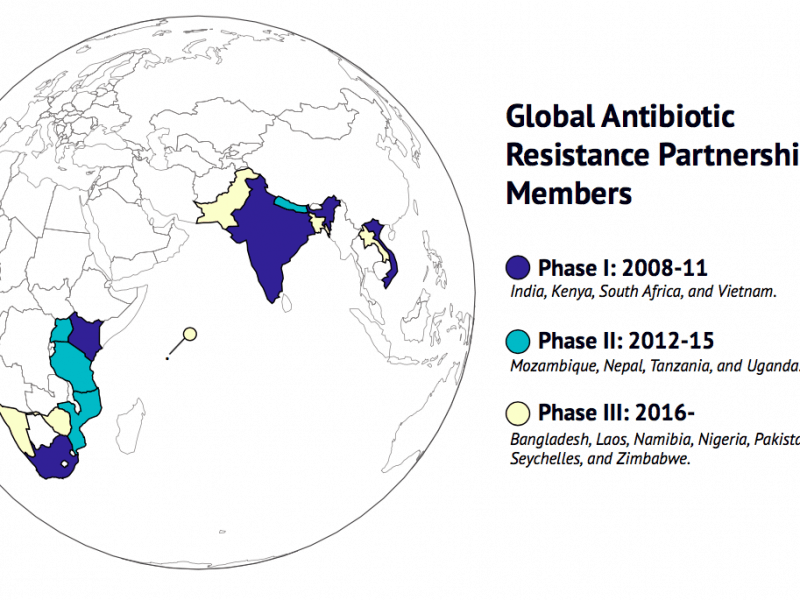
Despite evidence linking vaccination to AMR mitigation, funding for research at the clinical and policy levels is inadequate. One Health Trust, in collaboration with the World Health Organization, is initiating a global effort to increase awareness of the value of vaccines in mitigating AMR. The project spans the 15 countries in Africa and Asia that are in the Global Antibiotic Resistance Partnership (GARP), with the possibility to expand to other low- and middle-income countries. The GARP network is a major platform for communicating the role of vaccines.

Partners
- Yale
- Berkeley
- Imperial College London
- Princeton
Funding from the Bill & Melinda Gates Foundation and the Wellcome Trust.
Publications
- Hamilton A, Haghpanah F, Hasso-Agopsowicz M, Frost I, Lin G, Schueller E, Klein E, Laxminarayan R. (2023) Modeling of malaria vaccine effectiveness on disease burden and drug resistance in 42 African countries. Communications Medicine. Available here.
- Kumar CK, Sands K, Walsh TR, O’Brien S, Sharland M, Lewnard JA, Hu H, Srikantiah P, Laxminarayan R. (2023) Global, regional, and national estimates of the impact of a maternal Klebsiella pneumoniae vaccine: A Bayesian modeling analysis. PLoS Medicine. Available here.
- Heymann DL, Kieny MP, Laxminarayan R. (2022) Adding to the mantra: vaccines prevent illness and death, and preserve existing antibiotics. Available here.
- Lewnard JA, Fries LF, Cho I, Chen J, Laxminarayan R. (2022) Prevention of antimicrobial prescribing among infants following maternal vaccination against respiratory syncytial virus. Available here.
- Birger R, Antillón M, Bilcke J, Dolecek C, Dougan G, Pollard AJ, Neuzil KM, Frost I, Laxminarayan R, Pitzer VE. (2022) Estimating the effect of vaccination on antimicrobial-resistant typhoid fever in 73 countries supported by Gavi: a mathematical modelling study. Available here.
- Fu H, Lewnard JA, Frost I, Laxminarayan R, Arinaminpathy N. (2021) Modelling the global burden of drug-resistant tuberculosis avertable by a post-exposure vaccine. Available here.
- Vekemans J, Hasso-Agopsowicz M, Kang G, Hausdorff WP, Fiore A, Tayler E, Klemm EJ, Laxminarayan R, Srikantiah P, Friede M, Lipsitch M. (2021) Leveraging Vaccines to Reduce Antibiotic Use and Prevent Antimicrobial Resistance: A World Health Organization Action Framework. Available here.
- Andrejko K, Ratnasiri B, Hausdorff WP, Laxminarayan R, Lewnard JA. (2021) Antimicrobial resistance in paediatric Streptococcus pneumoniae isolates amid global implementation of pneumococcal conjugate vaccines: a systematic review and meta-regression analysis. Available here.
- Lewnard, J.A., Lo, N.C., Arinaminpathy, N., Frost, I, Laxminarayan, R., (2020) Childhood vaccines and antibiotic use in low- and middle-income countries. Available here.
- Klein EY, Schueller E, Tseng KK, Morgan DJ, Laxminarayan R, Nandi A. The Impact of Influenza Vaccination on Antibiotic Use in the United States, 2010-2017. (2020) Available here.
- Lewnard JA, Rogawski McQuade ET, Platts-Mills JA, Kotloff KL, Laxminarayan R. (2020) Incidence and etiology of clinically-attended, antibiotic-treated diarrhea among children under five years of age in low- and middle-income countries: Evidence from the Global Enteric Multicenter Study. (2020) Available here.